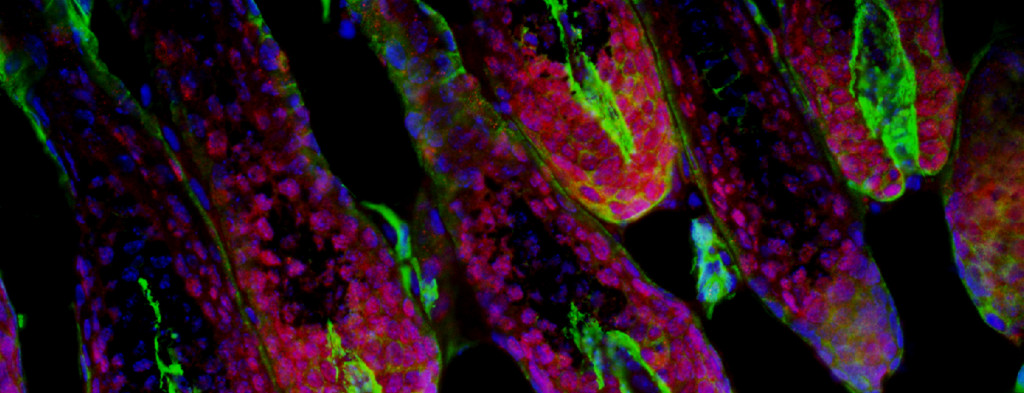
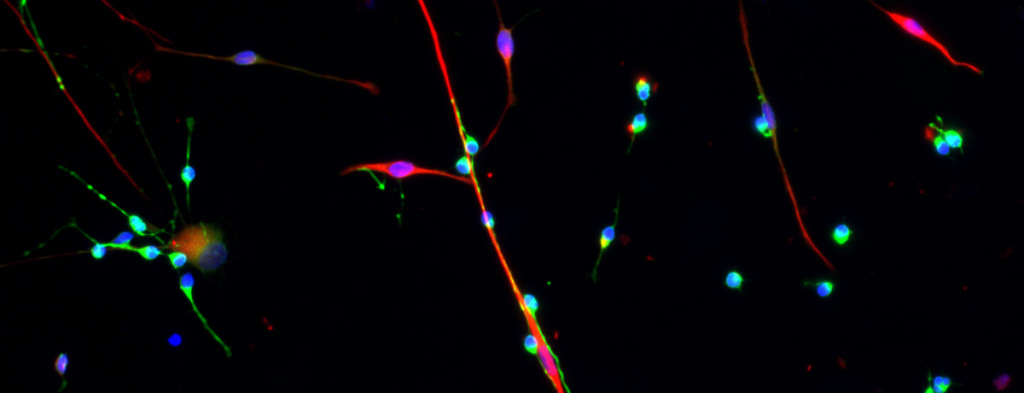
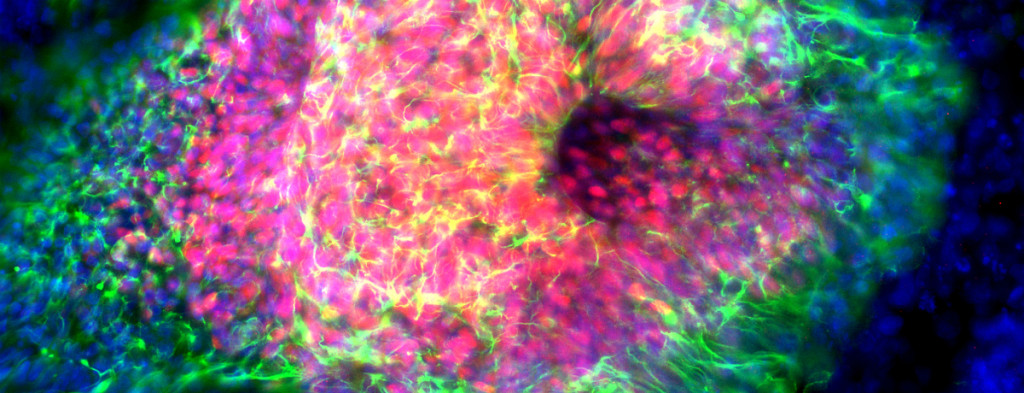
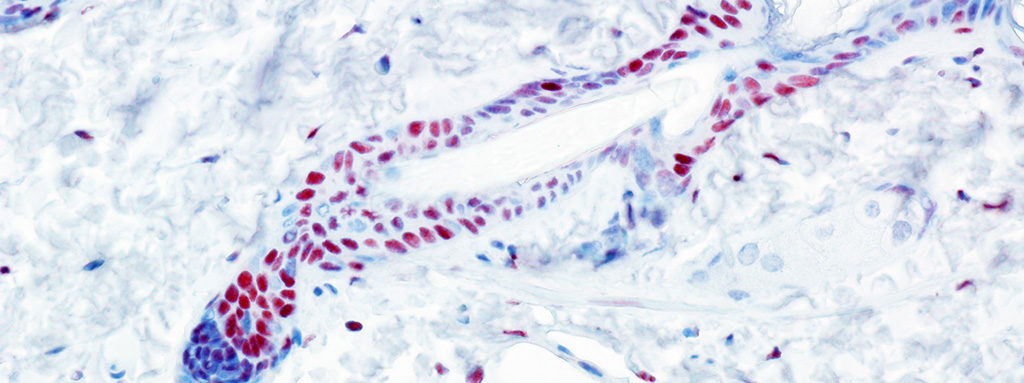
The process by which cell fates are adopted and maintained in organisms has preoccupied much of biological investigation for a century. We seek to uncover the mechanisms by which cells interpret signals during development or in mature tissue and generate a response leading to cell fate specification, regulation of self-renewal and differentiation, or even aberrant proliferation. The epidermis and nervous system are derivatives of progenitors specified in embryonic ectoderm. While the study of lower organisms has elucidated aspects of this specification, there is still a great deal unknown with regards to cell fate decisions in mammalian ectoderm, particularly human. We have devised an experimental paradigm that allows for the isolation and molecular characterization of cells in a model of human ectodermal development. We expect to find both familiar and unfamiliar pathways related to these specification events, and that some of these pathways will be used again to maintain stem cells in mature epidermis. We are using both in vitro and in vivo models to study epidermal adult stem cell self-renewal and differentiation to confirm this hypothesis and to uncover novel mechanisms. Some of these pathways that regulate self-renewal and differentiation are also thought to be defective in degenerative or dysplastic diseases. Using a murine model for tumorigenesis in the epidermis, we employ powerful new technology to describe the molecular landscape underlying tumor initiation by stem cells, including whether misregulation of self-renewal and differentiation pathways plays a role in cancer.
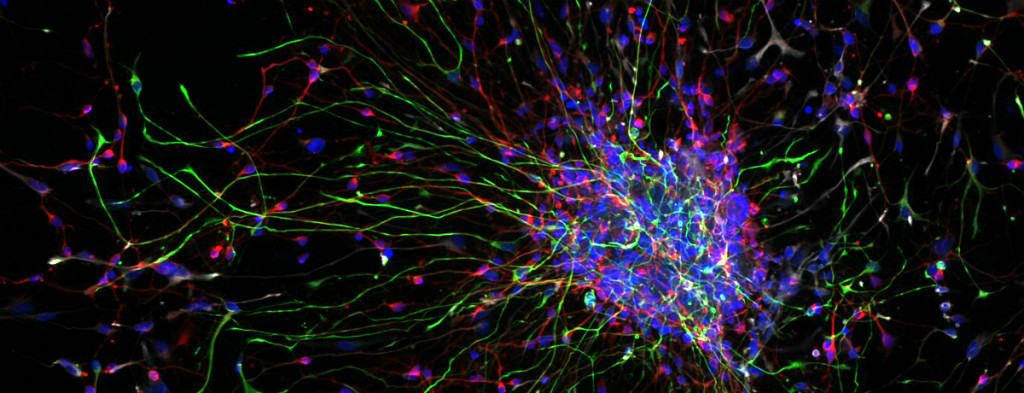
iPS cell-derived neural progenitor cells
Posted on